AB Scientific are excited to announce our new partnership with MeMed Diagnostics, to bring the MeMed Key and MeMed BV test to the UK diagnostic market. As part of our on-going commitment to improving sepsis diagnostics here in the UK, the MeMed products are a fantastic addition to our portfolio.
Bacterial and viral infections are often clinically indistinguishable. They can lead to inappropriate patient management and antibiotic misuse – adding a fast, near patient biomarker test we can help improve patient care.
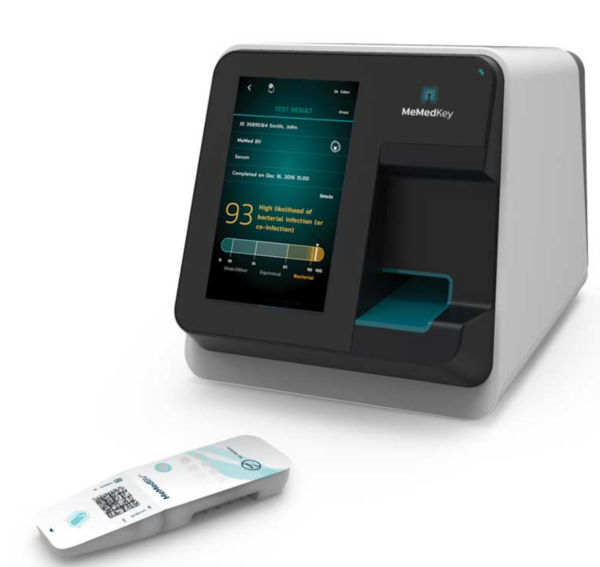
MeMed BV is the first FDA-cleared host-immune response assay for accurately distinguishing between bacterial and viral infections in just 15 minutes. From a small venous, whole blood sample, the MeMed BV computationally integrates the levels of three host immune proteins – TRAIL, IP-10 and CRP – into a simple score indicating the likelihood of a bacterial immune response or co-infection versus a likely viral immune response.
Samina Noyon, Commercial Director for UK/IRE at MeMed: “MeMed Diagnostics Ltd is excited to partner with AB Scientific supporting adoption of the MeMed BV test across the UK & Ireland. The synergy between the two companies focussed on providing the right diagnostic solution to establish infectious disease and help the fight against AMR will directly impact patient care at the point of need.”
Aaron Eglin, Rapid Diagnostics Sales Manager at AB Scientific: “The addition of the MeMed Key and BV test into our Rapid Dx portfolio is testament to our company mission to provide innovative products of the highest quality to help you achieve your goals and to make the world a healthier and safer place.
We believe that the MeMed BV test can improve sepsis diagnostics here in the UK and also reduce the burden of antimicrobial resistance by reducing the number of inappropriate treatments provided unnecessarily to patients.”